Case Study - Electrochemical Corrosion Failure Analysis
Electrochemical corrosion investigations are more complex but provide essential corrosion mechanism insight that is easier to understand than conventional investigations based on metallurgy and corrosion engineering,..
CASE STUDIES
John Fildes, Ph.D.

Key takeaways....
Actionable Insight for Litigators, Insurers, and Corporate Counsel
Dr. John Fildes has conducted electrochemical studies including ones for corrosion that span the beginning of his career at Borg Warner’s Corporate Research Center through his most recent research under funding by the Army.
Metallurgy and corrosion engineering providesguidance on the selections of metals to resist corrosion but does not explain why corrosion occurred in specific situations.
Dr. Fildes goes beyond conventional metallurgical and mechanical engineering corrosion studies by using the most modern, advanced electrochemical methods.
Dr. Fildes’ approach provides explicit corrosion mechanism insight such as quantifying the rate of corrosion, degree and rate of pitting of aluminum, and the amount of water uptake and degradation over time of paint and protective coatings.
Dr. Fildes’ approach overcomes the challenges of the low conductivity of oils and paints, and provides a reliable, non-destructive way to perform in situ corrosion testing such as on aircraft skin.
Dr. Fildes’ approach requires specialized and sophisticated chemical, mathematical, and analytical expertise and equipment with which he has extensive experience, but which remain behind the scenes in his reports while allowing him to state straightforward, clear, and compelling results and conclusions.
Introduction
An attorney rhetorically commented to Dr. Fildes during an aircraft corrosion inspection “can you really ever determine the cause of corrosion.” His rhetorical question reflects that too many corrosion investigations focus on a metal’s suitability and compatibility based on metallurgical and corrosion engineering guidelines. Another example, Dr. Fildes was brought late into a chemical process heater case because several experts agreed on the nature of the corrosion but failed to identify the cause, which Dr. Fildes identify as having nothing to do with the metal but with the way the chemical process was conducted. This conclusion was validated by a chemical analysis the plaintiff produced at Dr. Fildes’ deposition.
Failures in general and corrosion do not happen because codes, standards, or guidelines are violated, but because the underlying scientific principles are violated, For corrosion, these principles come from the physical chemistry domain of electrochemistry.
Basing corrosion investigations on electrochemistry, as Dr. Fildes does, can authoritatively answer why corrosion occurred. Dr. John Fildes has conducted electrochemical studies including ones for corrosion that span the beginning of his career at Borg Warner’s Corporate Research Center through his most recent research under funding by the Army.
This case study demonstrates a comprehensive approach based on electrochemical techniques to obtaining reliable corrosion rate and mechanism data for even difficult to measure systems such as lubricants and painted metals.
The Case For Using Electrochemistry
Conventional corrosion testing that exposes a sample to an environment with periodic inspection can screen corrosion resistance under a limited set of environmental conditions so this commonly used approach is limited for corrosion failure analysis because it may not adequately address the environment and its variation in the situation under investigation, it may not precisely identify the onset of corrosion, and it does not offer insight as to the mechanism by which a coating failed or corrosion occurred. Electrochemistry does all of these things.
There are several electro-chemical theories and techniques to measure corrosion rates. Sometimes the measurement techniques are complementary, and sometimes certain techniques are not applicable such as using dc methods for painted metals. Thus, meaningful application of electrochemistry to a specific problem requires experience and expertise, not just access to a commercial testing lab.
Electrochemistry may be complex, but fortunately, experts only need to convey in a compelling way the results of electrochemical investigations to attorneys, judges, and juries. The complex details are documented in an expert’s report, but otherwise can remain behind the scenes. This case study shows how use of electrochemistry makes the results more compelling without getting bogged down in complex electrochemistry principles. The next section describes what would form the presentation of an expert’s conclusions based on an electrochemical investigation. It is straightforward, substantial, data-driven, and clear without needing to focus on complex scientific principles. The section after the next one provides the complex details that stay behind the scenes.
This corrosion case study comes from research where the scientific details can be provided, which is not the case with most litigation-related investigation case studies. Thus, this case study demonstrates the approach and complexity Dr. Fildes’ typically encounters in his litigation-related investigations although each of those may involve different physical chemical theories, measurements, and analytical/mathematical methods.
Use of Complex Electrochemistry Provides The Simplicity of Authoritative Results
This investigation was of the corrosion resistance provided by two different types of protective coatings and how and why the corrosion resistance of each changed over time as the coatings aged and degraded during use.
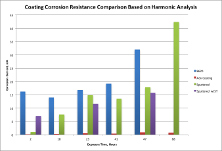
The measured corrosion rates for the bare steel and for this steel coated with the two different types of protective coatings are shown in the following Figure.
The corrosion rate was measured by three techniques: harmonic analysis (CASP), linear polarization (LP), electrochemical impedance spectroscopy (EIS). As stated above, there is often more than one electrochemical test method, and I generally use the commonly available techniques to make two or more determinations of the corrosion rate.
This way, people do not have to understand the measurements, they only have to see that the outcome of each established the same conclusion as is the case here. Although there is variation between absolute values of the three measurement techniques which is typical, there is good agreement in the trends for the techniques and the conclusions for the corrosion resistance of the specimens are the same for all of the technique, providing additional support for any conclusions.
The results show that the corrosion current (directly proportional to the corrosion rate) for bare steel is substantial throughout the test for the bare metal that involved exposure to 3.5 wt. % NaCl in water. This establishes a reference to which the corrosion resistance can be quantified. For example, he corrosion current for the sputtered coating starts much lower than for the bare metal, but substantially increases over time. The sputtered w/ST coating is the sputtered coating with a surface sealer and it performs somewhat better. The advanced coating, on the other hand, is far better. The corrosion current is extremely low through the test duration, showing excellent corrosion resistance and durability.
The protective efficiency, P%, of a coating is an alternative and equivalent way to convey these results. Protective efficiency is defined as:
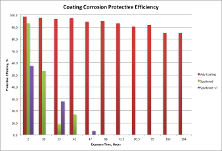
The next Figure shows the protective efficiencies of the coatings (the protective efficiencies are all relative to 2 hours exposure of bare steel). This may be an easier to understand presentation of the data than showing actual corrosion rates as was done in the Figure (above). This presentation makes it clear that the advanced coating provides excellent protection that is much better than the other two coatings.
Although the presentation so far is straightforward and clear, it does not explain why the coatings performed as they did. This context is important. People want to know it, and knowing that measurement results make sense in terms people can understand makes the results far more compelling. The next section describes how the corrosion mechanism was determined, but people do not need to know the “how,” they only need to know what is in this next paragraph, which explains why the advanced coating prevents corrosion while the sputtered coating doe not.
Although sputtered coatings can provide excellent corrosion protection, they can develop a columnar structure as they grow thicker during fabrication and this results in channels that extend from the coating surface to the metal, which allows corrosion to occur. The porosity of the sputtered coating (determined from electrochemical impedance spectroscopy) is 50% to 80%, which is why the sputtered coating examined here allows corrosion to occur. The porosity of the advanced coating is less than 5% throughout the test, which is why it is an excellent barrier that stops electrolyte from reaching the metal surface and this prevents corrosion from occurring.
This section shows that electrochemistry provides conclusions that are complete, straightforward, understandable, and compelling. The complexity of obtaining the “why” insight is now given in the next section, but it stays behind the scenes in a report. Attorneys, judges, juries, and even you do not need to understand or even read it.
Electrochemical Paint and Protective Coating Failure Analysis
Effective, easy to understand, and compelling corrosion investigations require determining not just the rate of corrosion in a corrosion screening test, but the rate of corrosion and its variation as environmental conditions vary, and the mechanism by which corrosion occurs in a specific environment. Both of these types of measurements are far more demanding (in theory, mathematics, analysis methods, test methods, and instrumentation) than a typical corrosion screening test such as salt spray exposure.
Electrochemical systems such as metals undergoing corrosion are composed of two simultaneous reactions, an oxidation reaction that for example consumes the metal in corrosion and a reduction reaction. The rates of these two reactions have to be equal at equilibrium, so the individual reaction rates cannot be measured unless a voltage is applied to move the system away from equilibrium. This is what an electrochemical measurement does in a controlled way.
There are many electrochemical corrosion measurement techniques. The simplest (in terms of theory, math, and equipment) are based on applying a steady-state voltage perturbation, but these techniques are not well suited for low conductivity situations such as oils and lubricants, or for painted metals. Dr. Fildes uses far more powerful electrochemical techniques such as electrochemical impedance spectroscopy (EIS) that is described in this case study.
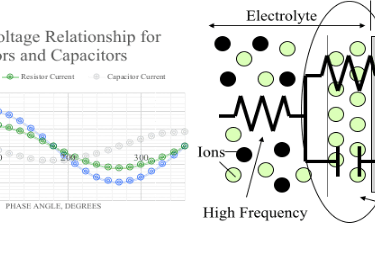
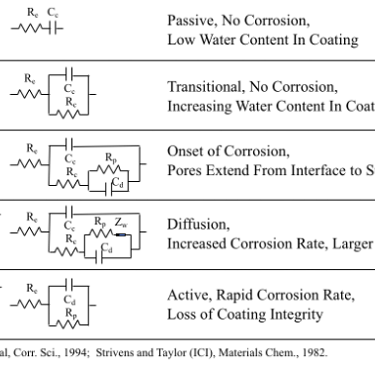
EIS involves applying an alternating voltage to a sample and this causes an alternating electric current to flow due to various electrochemical processes such as corrosion. The electrochemical processes can be modeled as an electric circuit of elements such as resistors and capacitors. The current that flows in response to the AC voltage stimulus changes in its magnitude and phase relative to the applied AC voltage as a coating takes up water and degrades, and as corrosion begins and advances.
The rate of chemical processes sets limits on the frequency that each process can follow, so the voltage-current data is collected over a wide range of frequencies, allowing the various processes to be measured independently. The voltage-current data is modeled as various electrical circuits of resistors, capacitors, and other elements, which is fit to the data using non-linear least squares. Each element of the circuit models a particular chemical process. For example, electrochemical impedance spectroscopy (EIS) can be used to follow the performance of paint coatings as their condition transits the various performance stages of aging and degradation as shown in the following diagram.
Although electrochemical impedance spectroscopy requires sophisticated instrumentation and complex mathematical analysis methods, if determines the cause of corrosion and paint coating failure in a way that is clear and understandable whereas other experimentally simpler corrosion testing methods provide no insight or ambiguous insight as to the reason for the failure. Since the electrochemical impedance spectroscopy result is quantitive, it can be presented in a simplified, but meaningful way to attorneys, judges, and juries. For example, the porosity of paint films is a valuable metric and has been determined from the values of circuit elements in the electrochemical impedance spectroscopy model by the formula:
where F % is the percentage porosity, Rp is the polarization resistance, Ecorr is the free (equilibrium) corrosion potential, and βa is the anodic Tafel coefficient.
Although the case study being described here involved coated metal, this electrochemical approach is equally valuable for bare metals. Corrosion is often non-uniform over a surface, for example pitting corrosion of aluminum. This electrochemical impedance spectroscopy based approach can quantify the degree of non-uniformity and how it changes over time, providing uniquely valuable insight to these types of corrosion processes and failures.
Electrochemical impedance spectroscopy also overcomes the difficult challenges encountered in systems with low electrical conductivity and allows the study of oils, lubricants, coolants, polymer melts, etc. For example, Dr. Fildes used electrochemical impedance spectroscopy to evaluate metals for plastic extruder barrels based on experiments conducted during extrusion.
Example - Electrochemical Corrosion Failure Mechanism of an Advanced Coating
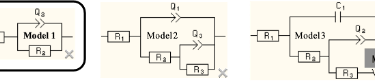
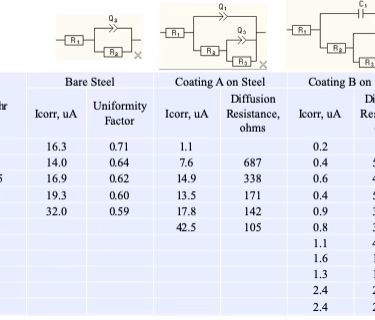
In this case study, Dr. Fildes examined coatings on 8620 steel. The results of electrochemical measurements for the bare metal and two coatings on that metal fit one of three electrical models shown in the next Figure.
Equivalent Circuit Model 1: Bare Metal
The first (left) Bare Metal Model is the simplest model and it fits the data from bare metals in salt solution. The model consists of a resistor, R1, that is in series with a parallel combination of another resistor R2 and a constant phase element, Q2. R1 represents the uncompensated resistance between the reference electrode and the working electrode (i.e. the metal specimen). R2 is the charge transfer resistance, which is inversely proportional to the flow of charge (i.e. current) at the solution – metal interface and thus R2 is inversely proportional to the corrosion rate. The final component is a capacitor, C2, or a non-linear element, Q2, that represents the double layer capacitance at the solution – metal interface.
The double layer capacitance can be represented by a conventional capacitance, C2, when the surface of the metal is uniform. The constant phase element, Q2, has to be used when the metal surface is non-uniform. Non-uniform corrosion causes a distribution of time constants over the corroding surface and modeling this requires the constant phase element. The impedance of the constant phase element, Q2, has the general form A(jw)-a and is characterized by two parameters. If the value of a is -1, then this non-linear impedance becomes linear and is equivalent to a capacitor whose value is A-1. If the value of a is 0, then this non-linear impedance again becomes linear and A is a pure resistance. Values for a between 0 and 1 represent a mixed, non-linear, element that is a transmission line. The value of this electric circuit element quantifies the variability of corrosion over the surface of the metal. Pitting corrosion of aluminum can be identified and quantified by the value of this electrical circuit element.
The other two electric circuit models are for the sputtered coating (far right model) and the advanced coating (middle model). They both contain more circuit elements than the bare metal model. These additional circuit elements are needed to model diffusion of the electrolyte through each coating to the metal surface. Significantly, different diffusion models are needed for the two different coatings and this is consistent with the fact that they have different physical structures. The columnar structure of the sputtered coating allow appreciable diffusion whereas the advanced coating does not allow appreciable diffusion. The change in the values of these circuit element over time quantify the increasing rate of diffusion due to the increasing aging degradation of the coatings. The circuit values change appreciably over time for the sputtered coating but not for the advanced coating. This clearly establishes that the advanced coating provides a far superior barrier to prevent corrosion, which is expected from its physical structure.
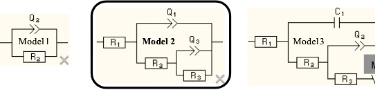
The interpretation of the model for the advanced coating will be presented here to provide insight as to how the interpretation is made (the sputtered coating interpretation details are not presented). The following Figure shows the model (middle, boxed) for an Insulating Barrier Coating.
Equivalent Circuit Model 2: Insulating Barrier Coating
R1 is the uncompensated solution resistance as defined above. C1 is the bulk capacitance of the coating, R2 is the resistance due to solution penetration into pores in the coating, Q2 is the double layer capacitance at the coating – 8620 interface (which is equivalent to Q2 in the Bare Metal Model above), and non-uniformity (as described above) requires a constant phase element instead of a conventional capacitor as described above. R3 is the charge transfer resistance that is inversely proportional to the corrosion rate and is thus equivalent to R2 in the Bare Metal Model. M3 is a very complex circuit element that models finite length diffusion of species through the coating.
The following table summarizes, in a simplified way, the corrosion resistance results of the electrochemical impedance study of the two coatings relative to the bare metal. Icorr is the corrosion current that is proportional to the rate of corrosion. The diffusion resistance is a metric of the effectiveness of the barrier (blocking) properties of the coating to the penetration of ions from the salt solution to the metal surface, which is what causes corrosion to occur.
Uncoated Steel has the simplest model (far left) and the highest corrosion rate. The uniformity factor characterizes if corrosion is uniform. A value of 1 is uniform, and lower values are increasingly non-uniform. The results establish that this steel does not corrode uniformly, which is consistent with this steel’s metallurgy.
Coating A is of a type of coating that builds up with a columnar structure. The model on the right shows that a circuit element representing a diffusion process is needed and this indicates that the columns form channels that allow water and sodium and chloride ions to reach the metal, which is represented by the value of the diffusion resistance. A larger diffusion resistance indicates less penetration of water and ions to the metal, which should result in a lower corrosion rate. This coating initially provides some corrosion protection, but by allowing access of water and ions to the metal, corrosion occurs, which destroys the integrity of the coating until the corrosion rate is similar to bare steel.
Coating B is a very thin, dense, and durable coating known to provide excellent corrosion protection. This coating provides excellent resistance to penetration of water and ions as indicated by very high diffusion resistance. This protects the metal from corrosion for a lengthy period of time, which is essential for a coating’s durability.
This conclusion is also shown by the porosity of each of the two coatings, which makes for a far simpler presentation of the results and conclusion. The metric ,F%, for porosity of a coating (described above in the last section) starts out around 1% and slowly increase to about 5% at 164 hours exposure (the end of the test) for the advanced coating whereas the columnar sputtered coating shows a porosity of 80% range at the beginning of the test, declining to 50% as the test progresses. This indicates the sputtered coating does not block the metal surface from exposure to the electrolyte, which allows corrosion to occur, and the corrosion products build-up in and block the pores of the coating as the test progresses.
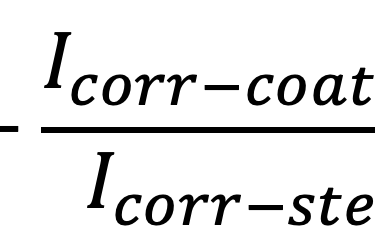
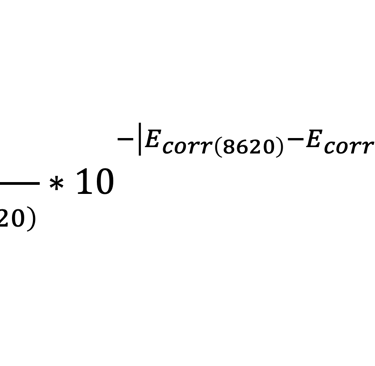
Bio for John Fildes, Ph.D.
Dr. Fildes is a doctoral scientist who has conceived, organized, and conducted $28 million of projects including R&D, litigation expert investigations, and collaborations involving Government labs, large defense companies, and leading universities.
Dr. Fildes was also CEO of an $18 million professional scientific/engineering consulting firm; president of a not-for-profit R&D institute; founder and leader of a $6 million scientific/engineering consulting firm; leader of a $3.5 million startup product design firm; leader of a $10 million contract research lab at Northwestern University; a senior professional in the $4.5 billion Borg-Warner Corporation Research Center.
Product Failures Expertise
Friction; Abrasive Wear, Adhesive Wear, Testing, Friction Measurement, Wear Prevention, Lubricants, Oil Quality Monitoring, Solid Lubricants, Hard Protective Coatings, Decorative Coatings, Paint, Electroplated Coatings, Corrosion, Electrochemical Corrosion Measurement, Ice Prevention; Gas Sensors, Carbon Monoxide Detectors; Product Design Procedures.
Materials & Process Expertise
Composites for Aviation, Buildings and Civil Construction: Thermoset and Thermoplastic Resins and Adhesives, Resin Transfer Molding, Autoclaving, Impedance Spectroscopy; Use of Composite Materials and Spray Foams Made On-Site In Construction; Roadway Chip Sealing, Water Treatment; Intelligent Process Control.
Chemistry & Chem Processes Expertise
Prediction Of Materials Properties, Stability, And Compatibility; Chemical Exposure; Chemical Process Equipment Failures.
Contact us
Whether you have a request, a query, or want to work with us, use the form below to get in touch with our team.
